Ticagrelor
Imvelaphi
I-Ticagrelor ngumchasi wenoveli we-P2Y12 receptor [1].
I-Ticagrelor iye yabikwa ukuba inqanda imiphumo ye-prothrombotic ye-ADP kwiplatelet ngokuchasene ne-P2Y12 receptor. I-Ticagrelor ibonise inhibition epheleleyo ye-platelet aggregation ex vivo. Ukongeza i-Ticagrelor iphakamise ukuvinjelwa kwe-dose ye-platelet aggregation emntwini. Ngaphandle kwezi, i-Ticagrelor nayo ibonise umchasi obophelelayo ngomlomo, ngokusebenzayo, ngokubuyisela umva. Ngokungafani nezinye i-inhibitors, i-Ticagrelor iphinde yabika ukuba inqanda i-P2Y12 i-receptor ngaphandle kokuguqulwa kwe-metabolic. Ngaphandle koko, i-Ticagrelor yi-agent yokuqala ye-thienopyridine ye-anti-platelet kwaye i-metabolized ikakhulu yi-CYP3A4 kunye ne-CYP2C19 [1] [2].
IiReferensi:
[1] Zhou D1, Andersson TB, Grimm SW. Uvavanyo lwe-in vitro yokusebenzisana kweziyobisi kunye ne-ticagrelor: i-cytochrome P450 reaction phenotyping, inhibition, induction, kunye ne-kinetics eyahlukileyo. Iziyobisi Metab Dispos. 2011 Epreli;39(4):703-10.
[2] Li Y1, Landqvist C, Grimm SW. I-Disposition kunye ne-metabolism ye-ticagrelor, i-novel P2Y12 i-receptor antagonist, kwiigundane, iigundane, kunye ne-marmosets. Iziyobisi Metab Dispos. 2011 Sep;39(9):1555-67. doi: 10.1124/dmd.111.039669. Epub 2011 Jun 13.
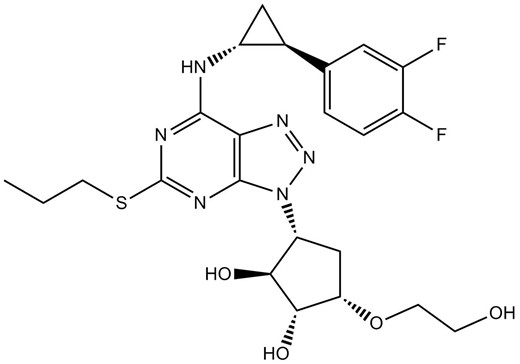
Inkcazo
I-Ticagrelor (AZD6140) i-reversible yomlomo ye-P2Y12 i-receptor antagonist yonyango lwe-platelet aggregation.
I-In vitro
I-Ticagrelor ikhuthaza ukuvinjelwa okukhulu kwe-adenosine 5"-diphosphate (ADP)-ukukhutshwa kwe-Ca2+ kwiiplatelet ze-ish vs abanye abachasi be-P2Y12R. Le mpembelelo eyongezelelweyo ye-ticagrelor ngaphaya kwe-P2Y12R i-antagonism iyingxenye njengesiphumo se-ticagrelor inhibiting i-nucleoside transporter elinganayo 1 (ENT1) kwiiplatelet, ezikhokelela ekuqokeleleni i-adenosine ye-extracellular kunye nokusebenza kwe-Gs-coupled adenosine A2A receptors [1]. Iiseli ze-B16-F10 zibonisa ukuncipha kwentsebenziswano kunye neeplatelet ezivela kwiigundane eziphathwe nge-ticagrelor xa kuthelekiswa neempuku eziphathwe nge-saline [2].
Kwimizekelo ye-B16-F10 ye-melanoma ye-intravenous kunye ne-intrasplenic metastasis, iigundane eziphathwe nge-dose yekliniki ye-ticagrelor (10 mg / kg) ibonisa ukunciphisa okuphawulweyo kwimiphunga (84%) kunye nesibindi (86%) i-metastases. Ngaphezu koko, unyango lwe-ticagrelor luphucula ukusinda xa kuthelekiswa nezilwanyana eziphathwe nge-saline. Umphumo ofanayo ubonwa kwi-4T1 imodeli yomhlaza wesifuba, kunye nokunciphisa imiphunga (55%) kunye nomnkantsha wethambo (87%) i-metastases elandela unyango lwe-ticagrelor [2]. Ukulawulwa komlomo omnye we-ticagrelor (1-10 mg / kg) kubangela i-dose-related inhibitory effect kwi-platelet aggregation. I-Ticagrelor, kwi-dose ephezulu (i-10 mg / kg) inqanda kakhulu i-platelet aggregation kwi-1 h emva kwe-dosing kunye ne-peak inhibition ibonwa kwi-4 h emva kwe-dosing.
Ugcino
4°C, ukukhusela ekukhanyeni, egcinwe phantsi kwe-nitrogen
*kwi-solvent : -80°C, iinyanga ezi-6; -20°C, inyanga eyi-1 (khusela ekukhanyeni, egcinwe phantsi kwenitrogen)
Ubume beMichiza





Isindululo18Iiprojekthi zoVavanyo loBulunga ezivunyiweyo4, kwaye6iiprojekthi ziphantsi kolwamkelo.

Inkqubo yolawulo lomgangatho wehlabathi ephucukileyo ibeke isiseko esiluqilima sokuthengisa.

Ukongamela umgangatho kuqhuba umjikelo wobomi bemveliso ukuqinisekisa umgangatho kunye nesiphumo sonyango.

Iqela leMicimbi yoLawulo lobuGcisa lixhasa iimfuno zomgangatho ngexesha lesicelo kunye nokubhaliswa.


Korea Countec Bottled Packaging Line


Taiwan CVC Bottled UkuPakisha Line


Italy CAM Board Packaging Line

Umatshini wokuQinisa iFette waseJamani

Japan Viswill Tablet Detector

I-DCS Control Room










